Genomics-Guided Off-Label Cancer Drug Use Shows Promise in Large Prospective Study
Evaluation by the Drug Rediscovery Protocol of off-label use of 37 approved cancer drugs on 1,610 patients showed modest activities overall, although subgroups of responders suggest that off-label drug use could be beneficial in tandem with more stringent bio…
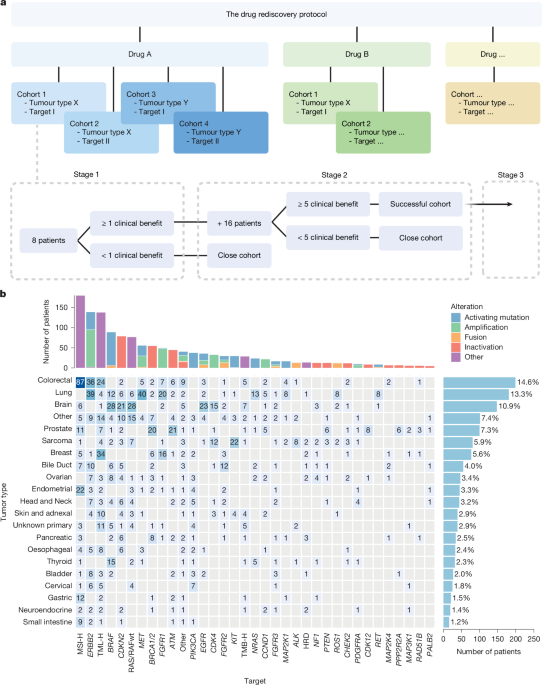
A major prospective evaluation of genomics-guided off-label cancer treatment has found modest overall activity across a broad patient population, but identified key subgroups where repurposed approved drugs demonstrated meaningful clinical benefit, according to findings from the Drug Rediscovery Protocol.
The study, one of the largest of its kind, enrolled 1,610 cancer patients and assessed 37 approved cancer drugs deployed outside their standard indications. Researchers used genomic profiling to match patients with treatments theoretically suited to their tumor biology, aiming to uncover new therapeutic avenues beyond conventional approvals.
While response rates across the entire cohort were modest, the data revealed distinct subgroups of responders whose outcomes suggest that off-label drug use, when paired with rigorous biomarker selection, could deliver genuine clinical value. These findings point to the critical importance of patient stratification in maximizing the benefits of drug repurposing.
Investigators emphasized that the results highlight both the potential and the limitations of broad genomics-guided treatment strategies. Without more stringent biomarker criteria to identify likely responders upfront, the overall signal from off-label prescribing can be diluted by patients unlikely to benefit from a given agent.
The Drug Rediscovery Protocol was designed precisely to generate prospective, real-world evidence on off-label oncology treatments — a category that accounts for a significant portion of cancer prescribing yet has historically lacked robust clinical data. This study represents a substantial step toward building that evidence base.
Experts in the field noted that refining biomarker frameworks will be essential to translating these findings into routine clinical practice. Better predictive tools could allow oncologists to move beyond trial-and-error prescribing and offer patients a more scientifically grounded rationale for off-label treatment choices.
The study's authors concluded that while genomics-guided off-label treatment is not yet ready for widespread adoption without further refinement, the identification of responding subgroups justifies continued investment in biomarker research and prospective evaluation of drug repurposing strategies in oncology.