Scientists Identify Rogue Immune Cells Driving Liver Disease and Age-Related Inflammation
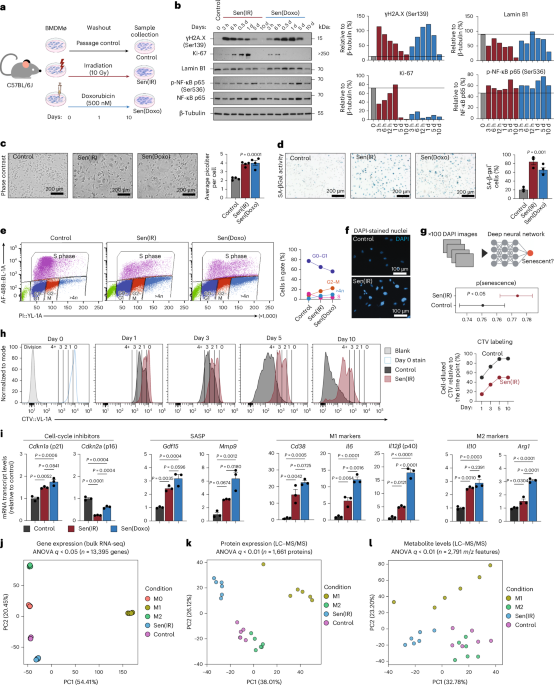
Salladay-Perez and colleagues explore senescence induction in human and mouse macrophages using multiomic profiling. They characterize a p21+TREM2+ senescent macrophage population, which they functionally implicate in inflammaging and liver disease.
Researchers have uncovered a distinct population of dysfunctional immune cells that may be fueling both age-related chronic inflammation and a increasingly common form of liver disease, according to a new study published by Salladay-Perez and colleagues.
The team used advanced multiomic profiling techniques to study how senescence — a state in which cells stop dividing but remain metabolically active and harmful — develops in macrophages, the immune cells responsible for clearing cellular debris and pathogens in human and mouse tissue.
Their analysis identified a previously undercharacterized macrophage population marked by the proteins p21 and TREM2. This specific subset displayed hallmarks of cellular senescence and was found to actively contribute to inflammaging, the low-grade chronic inflammation associated with aging that underpins a wide range of age-related diseases.
The researchers also functionally linked these senescent macrophages to metabolic dysfunction-associated steatotic liver disease, or MASLD, a condition characterized by fat accumulation in the liver that is closely tied to obesity, insulin resistance, and metabolic syndrome. MASLD affects hundreds of millions of people worldwide and can progress to cirrhosis or liver cancer if left untreated.
By combining genomic, transcriptomic, and epigenomic data, the team built a comprehensive molecular portrait of how these cells become senescent and why they promote tissue damage rather than repair. The findings suggest that the p21+TREM2+ macrophage population secretes inflammatory signals that worsen the local tissue environment — a phenomenon known as the senescence-associated secretory phenotype, or SASP.
The discovery opens potential new therapeutic avenues. Drugs known as senolytics, which selectively eliminate senescent cells, or senomorphics, which suppress their harmful secretions, could in theory be targeted to this macrophage population to slow liver disease progression and reduce systemic inflammaging.
Experts not involved in the study called the multiomic approach a significant methodological advance, noting that precisely characterizing senescent immune cell subtypes is essential for developing treatments that are both effective and tissue-specific. Further clinical validation will be needed to determine whether these findings translate to human therapeutic strategies.