Scientists Propose New Framework for How Cancer Cells Adapt to Drug Resistance
In this Perspective article, a theoretical framework for how the AP-1 family of transcription factors mediates cellular adaptation in cancer drug resistance is proposed.
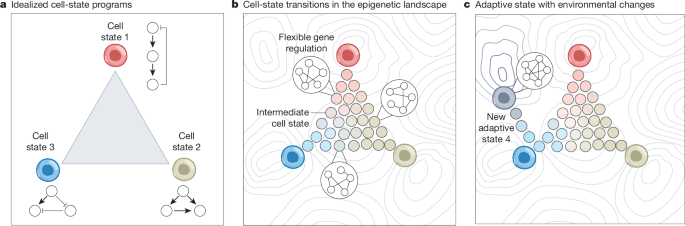
Researchers have put forward a new theoretical framework that could reshape our understanding of how cancer cells develop resistance to treatment, focusing on a family of proteins known as AP-1 transcription factors.
Published as a Perspective article, the framework outlines the mechanisms by which the AP-1 family of transcription factors mediates cellular adaptation, enabling cancer cells to survive and thrive even in the presence of therapeutic drugs.
Transcription factors are proteins that control the activity of genes, and the AP-1 family plays a central role in regulating cell growth, survival, and response to stress. In cancer, these proteins appear to be key orchestrators of the adaptive changes that allow tumors to evade treatment.
The authors propose that rather than relying on fixed genetic mutations alone, cancer cells exploit the flexibility of AP-1-driven gene regulation to dynamically rewire their internal signaling networks. This plasticity allows tumors to mount a rapid and effective response to drug pressure.
Understanding this adaptive mechanism has significant implications for oncology research and drug development. If cancer cells can be prevented from activating AP-1-mediated adaptation, it may be possible to overcome or delay the onset of drug resistance in patients.
The theoretical framework is expected to guide future experimental work, helping scientists identify specific AP-1 targets that could be blocked in combination with existing therapies. Researchers suggest this approach could prove particularly valuable in cancers known for high rates of treatment resistance, such as melanoma and lung cancer.
Experts in the field say the Perspective article offers a compelling conceptual lens through which to study one of oncology's most persistent challenges, and call for further empirical studies to validate and build upon the proposed model.