The Hidden Energy Behind Cold Sensitivity
Data from cryogenic electron microscopy combined with hydrogen–deuterium exchange mass spectrometry inform a mechanism for cold-evoked activation of the TRPM8 channel, providing a structural and energetic framework to explain cold sensitivity.
Structural energetics of cold sensitivity
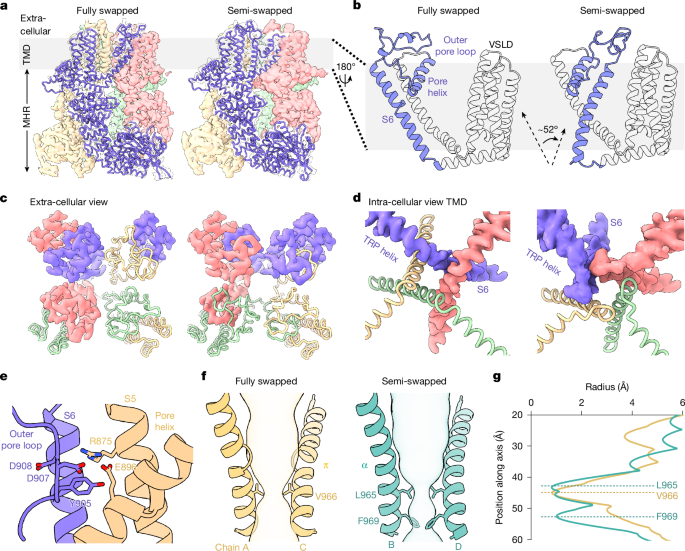
Scientists have made a significant breakthrough in understanding how the body senses cold temperatures, revealing the structural and energetic mechanisms behind the activation of the TRPM8 ion channel. This channel, found in sensory neurons, serves as the primary molecular sensor for cold and cooling sensations in mammals. By combining cutting-edge techniques including cryogenic electron microscopy and hydrogen-deuterium exchange mass spectrometry, researchers have constructed a detailed framework that explains how decreases in temperature trigger conformational changes in the channel protein, ultimately leading to its activation and the perception of cold.
The study leveraged cryogenic electron microscopy to capture high-resolution snapshots of the TRPM8 channel in multiple functional states, providing unprecedented views of the structural rearrangements that occur during cold-evoked activation. These structural data were complemented by hydrogen-deuterium exchange mass spectrometry, a powerful technique that maps changes in protein dynamics and solvent accessibility across different conditions. Together, these approaches allowed the research team to identify specific regions of the channel that undergo significant energetic and conformational shifts in response to cooling, pinpointing the molecular events that convert a thermal stimulus into an electrical signal.
Central to the findings is a mechanistic model that links temperature-dependent changes in protein stability and flexibility to the gating of the channel. The data suggest that cold temperatures destabilize specific structural domains within TRPM8, lowering the energetic barrier to channel opening. This thermodynamic perspective provides a unifying explanation for how a single protein can function as a sensitive and reliable temperature detector, integrating both structural transitions and energetic landscapes to produce a graded response to cooling.
The implications of this research extend beyond basic neuroscience, offering potential avenues for therapeutic intervention in conditions involving cold hypersensitivity, chronic pain, and sensory disorders. By establishing a comprehensive structural and energetic framework for cold sensitivity, the study sets the stage for the rational design of drugs that could modulate TRPM8 activity with precision. Researchers anticipate that this approach may also shed light on the mechanisms governing other temperature-sensitive ion channels, broadening our understanding of thermosensation across biological systems.